T. David Burleigh, PhD & PE
Dr. T. David Burleigh is a Professor of Materials and Metallurgical Engineering at New Mexico Tech. He is specializes in the corrosion of metals and in metallurgical failure analysis. Dr. Burleigh has taught a variety of courses at New Mexico Tech, and the most recent ones are listed above.
In his corrosion research laboratory, students work on several corrosion research projects, including the corrosion of copper alloys and the anodization of steel. Dr. Burleigh is a certified "Corrosion Specialist" by NACE International, and a registered "Professional Engineer in Metallurgy," in New Mexico. He completed his Ph.D. in Metallurgy at M.I.T. in 1985 conducting research in the Uhlig Corrosion Lab. He continued research in photoelectrochemistry and passivity at the Fritz-Haber-Institut (Max-Planck-Institute for Physical Chemistry) in West Berlin, West Germany. Dr. Burleigh worked in the corrosion group at Alcoa Technical Center for over five years designing alloys, corrosion tests, and testing products.
Since 1993 he has been the principal investigator for Burleigh Corrosion Consultants, where he solves corrosion problems for small and large businesses. Prior to coming to New Mexico Tech, he was a Research Associate Professor at the Materials Science and Engineering Dept. at the University of Pittsburgh where he taught and also conducted research in aqueous corrosion.
"Top 5 Trends in Materials Engineering" by Michael Abrams (May 2013) ASME.org. See last paragraph under "5. Spinning Smoke." www.asme.org/engineering-topics/articles/manufacturing-processing/top-5-trends-materials-engineering
 "Brown Nails" by Amanda Kuker, Sara Waters and Kat Mireles, of the New Mexico Tech
Materials Advantage Chapter. This new hit song describes the pain and the heartbreak
of corrosion. In June 2011, the ASM Foundation selected this song to be a Materials
Radio Podcast (search for "Brown Nails"), or you can listen to it here.
"Brown Nails" by Amanda Kuker, Sara Waters and Kat Mireles, of the New Mexico Tech
Materials Advantage Chapter. This new hit song describes the pain and the heartbreak
of corrosion. In June 2011, the ASM Foundation selected this song to be a Materials
Radio Podcast (search for "Brown Nails"), or you can listen to it here.
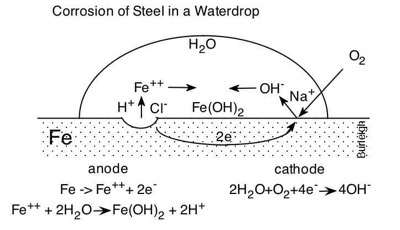 "Corrosion of Steel in a Waterdrop" may been seen at http://www.youtube.com/watch?v=d0LUB90uDaEm
"Corrosion of Steel in a Waterdrop" may been seen at http://www.youtube.com/watch?v=d0LUB90uDaEm
This video is a series of time-lapse micrographs taken by Dr. Burleigh in 2011. These timed micrographs show a water drop containing pH indicator solution, sitting on the surface of bare steel. The water drop is Socorro municipal water, containing many dissolved minerals including 14 ppm NaCl. As time progresses, the color of the pH indicator solution changes, indicating that acidic and caustic regions are developing in the water drop. Dr. Burleigh narrates the micrographs and explains the corrosion phenomena witnessed in the video.
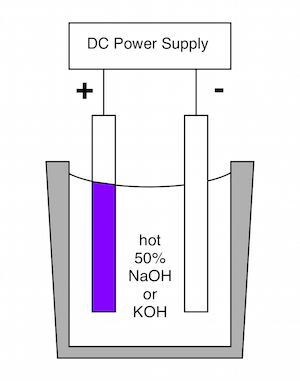 "How to Anodize Steel" may been seen at https://www.youtube.com/watch?v=7g-azzYnMYo
"How to Anodize Steel" may been seen at https://www.youtube.com/watch?v=7g-azzYnMYo
This video describes the methods developed by Prof. Burleigh and his students for
anodizing steel in hot caustic electrolytes. This process is the subject of two publications.
The anodized layer is a nano-porous magnetite film that is adherent to the surface.
The reflection of light from the top and bottom surfaces of the oxide film create
a dichromic color that changes with viewing angle.
T.D. Burleigh, P. Schmuki and S. Virtanen, "Properties of the Nanoporous Anodic Oxide Electrochemically Grown on Steel in Hot 50% NaOH." Journal of the Electrochemical Society, (Jan 2009), 156, 1, C45-C53. JECS_2009
T.D. Burleigh, T.C. Dotson, K.T. Dotson, S.J. Gabay, T. Sloan, S.G. Ferrell, "Anodizing Steel in KOH and NaOH Solutions," Journal of the Electrochemical Society (Oct. 2007), 154, 10, 579-586. JECS_2007
Related website: www.steelanodize.corrosionhelp.com
 "Corrosion of Aluminum Alloys" may be seen at
"Corrosion of Aluminum Alloys" may be seen at
http://youtu.be/YeFTEzeX56k
This educational video (produced by Dr. Burleigh in 2014) is a brief introduction
into the corrosion of aluminum alloys. The video shows the common forms and the theoretical
mechanisms of aluminum corrosion. This video was funded by a grant from Modern Light
Metals.
 "Demon Chlorides attack Castle Steel"
"Demon Chlorides attack Castle Steel"
One of Dr. Burleigh's students wrote this cartoon explanation of the role of nitrogen and molybdenum in preventing pitting corrosion of stainless steel. He currently wishes to remain anonymous. This cartoon is based on the cartoon presented by Prof. Koji Hashimoto in 1983 at The Fifth International Symposium on Passivity in Bombannes, France. Prof. Hashimoto first introduced the demon chlorides and the good mother Molly. This cartoon has added the characters, nanny nitrogen, cousin carbon, and harlot hydrogen. You may view "Demon Chlorides attack Castle Steel" at http://www.youtube.com/watch?v=KpCX-8Z1xsI
Figures from some of Dr. Burleigh's publications

Microbes inside a corrosion pit in copper, from Thomas D. Burleigh, Casey G. Gierke, Narjes Fredj and Penelope J. Boston, "Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion," Materials (2014), 7, p. 4321-4334. http://www.mdpi.com/1996-1944/7/6/4321
 From J.C. Garcia and T.D. Burleigh, "Beginnings of Gold Electroplating," The Electrochemical
Society Interface (Summer 2013), 22, 2, 36-38. (electroplating.pdf)
From J.C. Garcia and T.D. Burleigh, "Beginnings of Gold Electroplating," The Electrochemical
Society Interface (Summer 2013), 22, 2, 36-38. (electroplating.pdf)
 Multi-colored Cu2O films deposited on copper. From N. Fredj and T.D. Burleigh, "Transpassive
Dissolution of Copper, and Rapid Formation of Brilliant Colored Copper Oxide Films,"
Journal of the Electrochemical Society (2011), 58, 4, C104-C110. (Fredj-Burleigh-2011.pdf)
Multi-colored Cu2O films deposited on copper. From N. Fredj and T.D. Burleigh, "Transpassive
Dissolution of Copper, and Rapid Formation of Brilliant Colored Copper Oxide Films,"
Journal of the Electrochemical Society (2011), 58, 4, C104-C110. (Fredj-Burleigh-2011.pdf)
 1010 steel sheet anodized in one minute steps. From Burleigh et al, "Anodizing Steel
in KOH and NaOH Solutions," Journal of the Electrochemical Society, 154, 10, p. C579-586
(JECS_2007.pdf). For more information, see also www.steelanodize.com and JECS_2009.pdf.
1010 steel sheet anodized in one minute steps. From Burleigh et al, "Anodizing Steel
in KOH and NaOH Solutions," Journal of the Electrochemical Society, 154, 10, p. C579-586
(JECS_2007.pdf). For more information, see also www.steelanodize.com and JECS_2009.pdf.
 Eighteen tuning forks machined from seventeen different alloys and one polymer, are
used as classroom demos. Each fork has its own unique resonant pitch and harmonics,
dampening, weight, color and stiffnesss. A two-page article has been published entitled,
"Tuning Forks for Vibrant Teaching." Journal of Metals (2005), 57, 11, 26-27. (TuningJOM2005.pdf) It may aslo be found on the Journal of Metals website.
Eighteen tuning forks machined from seventeen different alloys and one polymer, are
used as classroom demos. Each fork has its own unique resonant pitch and harmonics,
dampening, weight, color and stiffnesss. A two-page article has been published entitled,
"Tuning Forks for Vibrant Teaching." Journal of Metals (2005), 57, 11, 26-27. (TuningJOM2005.pdf) It may aslo be found on the Journal of Metals website.
 The polished, etched and heat-tinted face of the Gibeon Meteorite shows a crystal
pattern denoting a cooling rate of 1 C per million years (from the Materials and Metallurgical
Engineering Department Brochure).
The polished, etched and heat-tinted face of the Gibeon Meteorite shows a crystal
pattern denoting a cooling rate of 1 C per million years (from the Materials and Metallurgical
Engineering Department Brochure).
 A schematic of the photoelectrochemical apparatus used for measuring photocurrents
and photovoltages on metals immersed in a liquid. J.R. Birch and T.D. Burleigh, "Oxides
Formed on Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing," Corrosion
(2000), 56, 12, 1233-1241. (BirchBurleigh2000.pdf)
A schematic of the photoelectrochemical apparatus used for measuring photocurrents
and photovoltages on metals immersed in a liquid. J.R. Birch and T.D. Burleigh, "Oxides
Formed on Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing," Corrosion
(2000), 56, 12, 1233-1241. (BirchBurleigh2000.pdf)
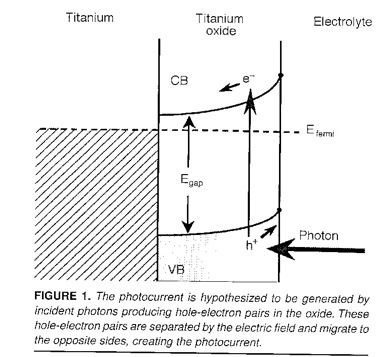 The photocurrents are result from light exciting electrons in the oxide film, in the
presence of a Schottky barrier. The electrons are excited from the valence band (V.B.)
to the conduction band (C.B.) where they flow down hill under the influence of the
electric field. The electric field is a result of the mismatch of the Fermi levels
of the electrolyte and the metal. J.R. Birch and T.D. Burleigh, "Oxides Formed on
Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing," Corrosion (2000),
56, 12, 1233-1241. (BirchBurleigh2000.pdf)
The photocurrents are result from light exciting electrons in the oxide film, in the
presence of a Schottky barrier. The electrons are excited from the valence band (V.B.)
to the conduction band (C.B.) where they flow down hill under the influence of the
electric field. The electric field is a result of the mismatch of the Fermi levels
of the electrolyte and the metal. J.R. Birch and T.D. Burleigh, "Oxides Formed on
Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing," Corrosion (2000),
56, 12, 1233-1241. (BirchBurleigh2000.pdf)
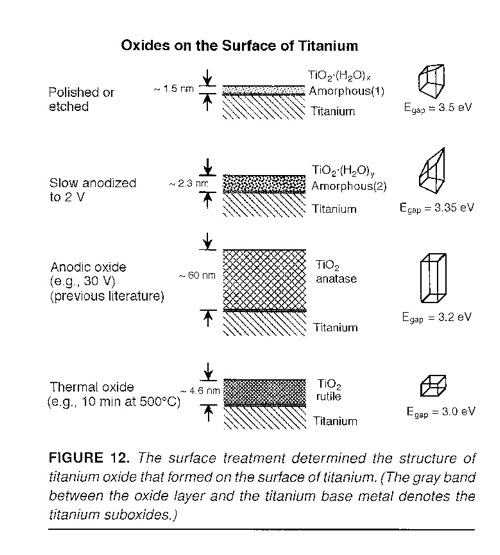 The photocurrents may be used to identify the crystal structure of the titanium oxide
since the different oxides have different bandgaps. J.R. Birch and T.D. Burleigh,
"Oxides Formed on Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing,"
Corrosion (2000), 56, 12, 1233-1241. (BirchBurleigh2000.pdf)
The photocurrents may be used to identify the crystal structure of the titanium oxide
since the different oxides have different bandgaps. J.R. Birch and T.D. Burleigh,
"Oxides Formed on Titanium by Polishing, Etching, Anodizing, or Thermal Oxidizing,"
Corrosion (2000), 56, 12, 1233-1241. (BirchBurleigh2000.pdf)
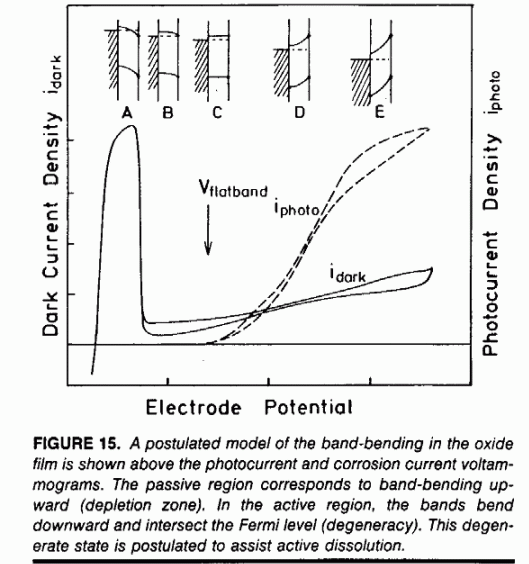
The active-passive transition of may be modeled as a semiconductor film that becomes degenerate at high or low potentials. During degeneracy, the conduction or valence bands bend across the Fermi level and the oxide becomes an electric conductor. from T.D. Burleigh, "Anodic Photocurrents and Corrosion Currents on Passive and Active-Passive Metals," Corrosion (1989), 45, 6, 464-471 (Corrosion1989.pdf)
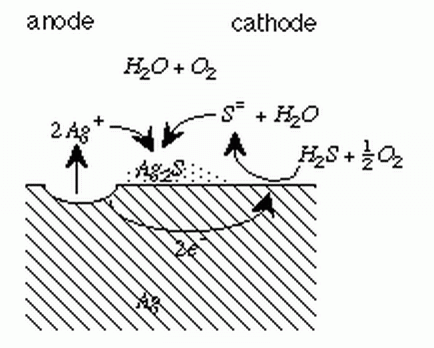 The tarnishing of silver requires an atmosphere containing hydrogen sulfide, oxygen
and water vapor. A electrochemical mechanism is proposed for the tarnishing of silver.
T.D. Burleigh, Y. Gu, G. Donahey, M. Vida, D.H. Waldeck, �Tarnish Protection of Silver
using a Hexadecanethiol Self-Assembled Monolayer and Descriptions of Accelerated Tarnish
Tests,� Corrosion (2001), 57, 12, 1066-1074. (BurleighWaldeck2001.pdf)
The tarnishing of silver requires an atmosphere containing hydrogen sulfide, oxygen
and water vapor. A electrochemical mechanism is proposed for the tarnishing of silver.
T.D. Burleigh, Y. Gu, G. Donahey, M. Vida, D.H. Waldeck, �Tarnish Protection of Silver
using a Hexadecanethiol Self-Assembled Monolayer and Descriptions of Accelerated Tarnish
Tests,� Corrosion (2001), 57, 12, 1066-1074. (BurleighWaldeck2001.pdf)
A corrosion chimney forms above the corroding pit on aluminum corroding in saltwater. T.D. Burleigh, E. Ludwiczak, and R.A. Petri, "Intergranular Corrosion of an Al-Mg-Si-Cu Alloy," Corrosion (1995), 51, 1, 50-55. (Corrosion1995.pdf)
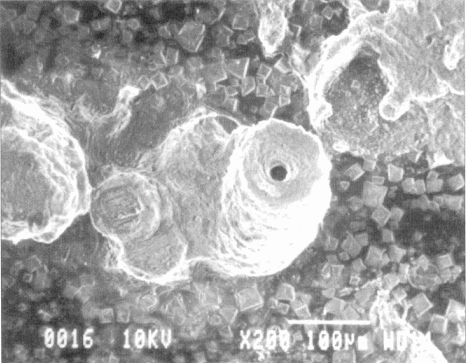
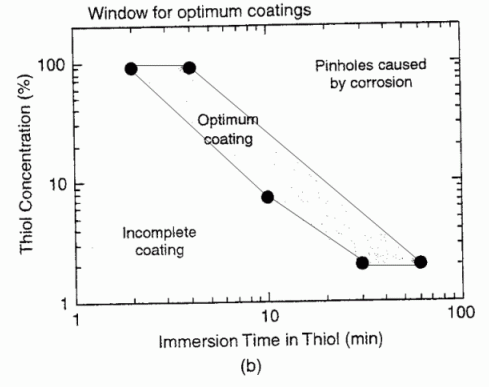 Silver may be protected from tarnishing by a self-assembled monolayer (SAM) of hexadecanethiol.
The SAM is prepared by cleaning, etching, rinsing, then immersion in a thiol solution
for a certain time period. Too short of time leads to an incomplete film, and too
long of time leads to pinhole corrosion. Figure 10b from T.D. Burleigh, Y. Gu, G.
Donahey, M. Vida, D.H. Waldeck, Tarnish Protection of Silver using a Hexadecanethiol
Self-Assembled Monolayer and Descriptions of Accelerated Tarnish Tests, Corrosion
(2001), 57, 12, 1066-1074. (BurleighWaldeck2001.pdf)
Silver may be protected from tarnishing by a self-assembled monolayer (SAM) of hexadecanethiol.
The SAM is prepared by cleaning, etching, rinsing, then immersion in a thiol solution
for a certain time period. Too short of time leads to an incomplete film, and too
long of time leads to pinhole corrosion. Figure 10b from T.D. Burleigh, Y. Gu, G.
Donahey, M. Vida, D.H. Waldeck, Tarnish Protection of Silver using a Hexadecanethiol
Self-Assembled Monolayer and Descriptions of Accelerated Tarnish Tests, Corrosion
(2001), 57, 12, 1066-1074. (BurleighWaldeck2001.pdf)
(Image Available Here) Zinc corrodes faster under UV illumination. E.A. Thompson and T.D. Burleigh, "Accelerated Corrosion of Zinc Alloys Exposed to Ultraviolet Light," Corrosion Engineering, Science and Technology (2007), 42, 3, p. 237-241. (Zinc&UV_2007.pdf)
(Graph Available Here) A corrosion resistant Mg-Li alloy is made by alloying with scandium. T.D. Burleigh, R.K. Wyss, "Dual Phase Magnesium Based Alloy having Improved Properties," U.S. Patent No. 5,059,390 (October 22, 1991). (MgLiSc.pdf)
(Graph Available Here) Improvement in the erosion corrosion resistance of Cu-10%Ni was achieved by adding indium. T.D. Burleigh and D.H. Waldeck, "Effect of Alloying on the Resistance of Cu-10% Ni Alloys to Seawater Impingement," Corrosion (1999), 55, 8, 800-804. (Corrosion1999.pdf)